El company i coeditor de Catquímica Marcel Swart em fa assabentar d’una interessant notícia, publicada abans d’ahir al web de la Royal Society of Chemistry. S’ha sintetitzar una molècula d’alcà amb l’enllaç C-C més llarg.
Tal i com ens expliquen a l’entrada del web de la RSC:
The feat was achieved by subtly balancing forces of attraction and repulsion in a carefully designed molecule based on carbon configured in a diamond-like structure. The study demonstrates the importance of weakly attractive intramolecular forces and how they can be harnessed to introduce particular features into molecules.
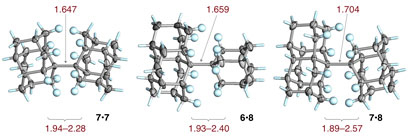
Tal i com apreciem als models,
The researchers constructed dumbell-shaped molecules consisting of a central C-C ‘bar’ on each end of which were three-dimensional diamond-like alkanes, or ‘diamondoids’. Crucially, the outer surfaces of these diamondoids are capped by hydrogens. The repulsive forces of each diamondoid on either side of the central C-C bridge are sufficient to stretch the bond to more than 1.7Å, compared with a typical alkane C-C bond of around 1.5Å, an atomically significant increase. However, the van der Waals attraction between the hydrogens on either side of the central bond is sufficient to hold the molecule together. Indeed, it is a highly stable solid.
Per a més informació, llegir la notícia al web de la RSC: http://www.rsc.org/chemistryworld/News/2011/September/14091101.asp






Posted on 16/09/2011 per Pep Anton Vieta
0